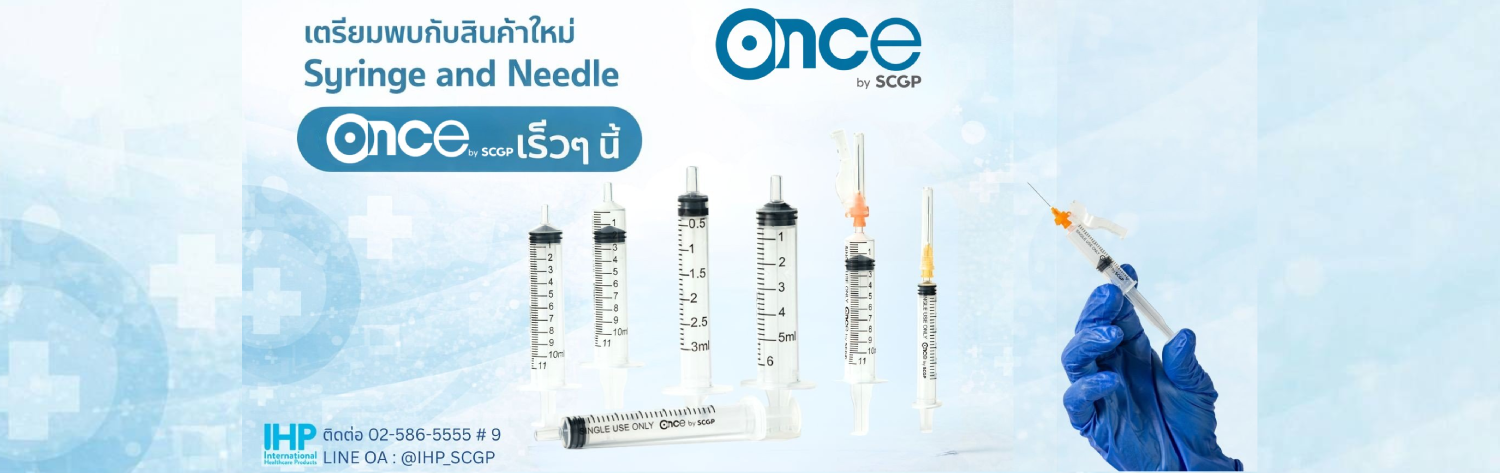
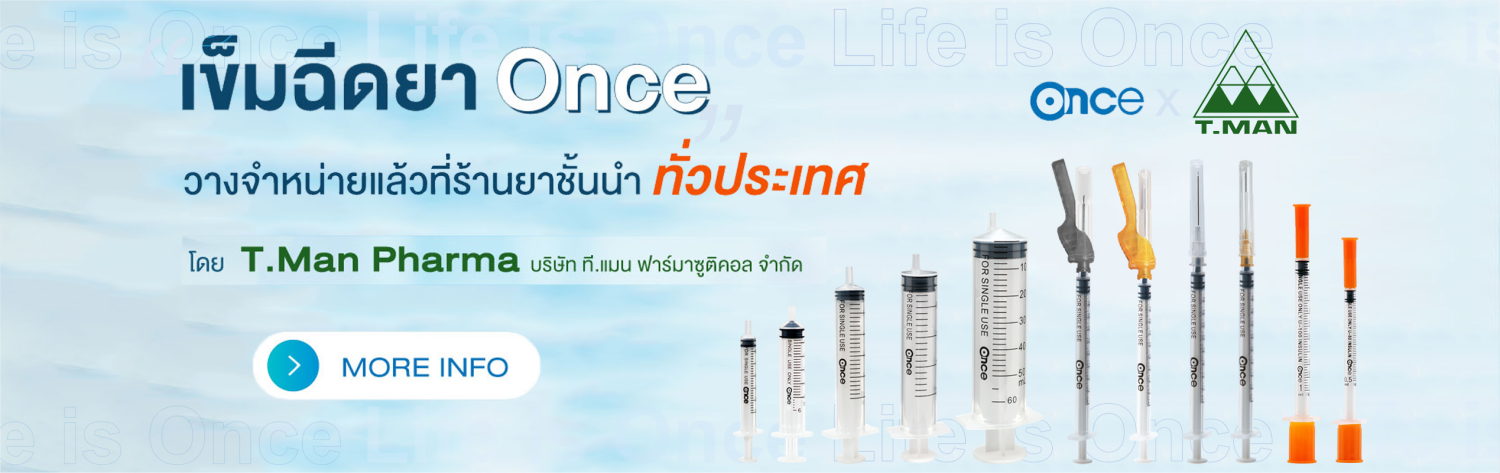
ONCE MEDICAL
ONCE Medical was established in June 2003 as a subsidiary firm of ONCE Group with support from the government. The purpose of this company is to deliver advanced new technologies in medical devices and pharmaceutical products, especially from Thai professional inventors in order to improve people’s health quality, mainly in Thailand, CLMV and we have the vision to distribute our “ONCE” products over the world.
WE ARE
ONCE MEDICAL

Leadership

Solutions
Tailor-made to serve all partners’ satisfaction.

Support
Strong network & support from Thai government, business partnerships and especially our CUSTOMERs

ONE
With one well step and strategist planning before every move.
NEW
CREATIVE
Enthusiasm and passion.
EXCELLENCE
In use with easy practice.
OUR CORE BRAND VALUES

Tortrakul Wattanavorakijkul, Ph.D.
Chief Strategy Officer of Once Medical Corp.
tortrakul@oncemedicaldevice.com
Tortrakul is a distinguished professional in the field of sustainability, with a solid foundation in engineering and environmental economics. Driven by a passion for addressing global challenges, He pursued a Ph.D. in environmental economics, focusing on innovative solutions and policies that promote long-term balance between social, ecological and economic viability.
Beyond academia, he gained invaluable insights into business operation and development, with a wealth of experience in both strategic planning and entrepreneurship. Now, he brings the expertise to the medical supplies industry as the CSO of our esteemed company. Drawing on his vast knowledge of sustainability and environmental economics, he leads our team in creating innovative and eco-friendly solutions for the healthcare sector.
With a background of Chemistry, Parivat began his career working with medical device multinational companies and turned to be as Manager and Lead Auditor experience with 1 biggest Notified Body/Certification Body, he was successful to qualify as ISO 13485, ISO 9001, CE (MDD) and MDSAP Lead Auditor (in more than 500 audits with more than 1,000 days on-site), Technical Documentation Assessor, Technical Certifier and Trainer for related courses. His knowledge of GMP, ISO 13485, EU Medical Device Regulation (MDR 2017/745), Quality System Regulation (21 CFR Part 820) brings up opportunity to implementation of regulatory/management system to several companies in both Thailand and overseas.

Parivat Akarapimarn
Chief Regulatory Officer of Once Medical Corp.
parivat@oncemedicaldevice.com
Krit co-founded Once Medical Holding, and currently serves as a Chief Operating Officer (COO) of the company. He will be responsible for the operational plan and execution of the company.
With more than 30 years of experience in pharmaceutical and medical device industries from the large scaled multinational companies, and one of the leading distributors, Krit has brought his wealth of knowledge to the company. His skills include sales force management, product portfolio strategy & sales execution, partner/supplier management, budget management, etc.
Krit earned an MBA from Srinakharinwirot University, and a Bachelor of Science in Pharmacy from Chulalongkorn University.

Krit Termphiriyakij
Chief Operating Officer of Once Medical Corp.
krit@oncemedicaldevice.com

Nuttawut Laksanapanyakul
Senior Engineering Officer of Once Medical Corp.
nuttawut@oncemedicaldevice.com
During 20 years as a policy researcher for several internationally-recognized think tanks in the field of regional economic integration, industrial development and research, development and innovation activities, Nuttawut has learnt a lot about the current state of development level of many countries around the world including Thailand.
One of the major challenges hindering Thailand and other developing countries from escaping ‘the middle-income trap’, which is the situation that the country fails to become a developed and rich country, is the lack of technological advancement in the manufacturing sector. Nonetheless, we have seen more and more success stories of Thai firms in developing their own technology and innovation activities.
With the company’s non-stop devotion towards practical innovation for greater customers’ satisfaction and his expertise in strategic planning and execution, he believes that the success of ONCE MEDICAL’s innovative products and services is certainly another jigsaw that when combining with others will be a thrust for the country to be out of the middle-income trap.

ONCE MEDICAL TEAM

Karan Tejasen
CEO

Dr. Sirinporn Thamapipol
Sales & Export Manager - Thailand

Dr. Puttisak Sattayaprasert
Consultant R&D

Krit Termphiriyaki
Consultant
We are always by your side.
CONTACT US NOW!
OUR CLIENT & SUPPORTER